Microneedle Array Patch (MAP) Commercialisation
Quantitative, in vivo insight for microneedle array patch development

Non-invasive OCT imaging to accelerate MAP research, optimisation and commercialisation
VivoSight Dx Pro gives you real-time, in vivo quantification of microneedle puncture performance and skin response — without tissue sectioning.

Accelerate MAP research, optimisation and commercialisation
VivoSight Dx Pro enables researchers and developers to visualise, measure and quantify microneedle–skin interactions in vivo and ex vivo, providing robust evidence to accelerate MAP design, optimisation and regulatory pathways.
Using high-resolution Optical Coherence Tomography (OCT), VivoSight delivers rapid, non-invasive and repeatable insight into microneedle puncture performance and skin response — without the time, cost or artefacts associated with tissue sectioning.
“VivoSight is an indispensable tool to advance and optimise MAP research and product development.”
Professor Ryan Donnelly, Queen’s University Belfast, UK

Visualise microneedle insertion in real time
VivoSight Dx Pro provides unparalleled real-time, in vivo imaging of microneedle arrays during and after application.
Researchers can directly observe how needles interact with skin, how deeply they penetrate, and how they evolve over time.
Key capabilities include:
- Real-time visualisation of microneedle insertion depth
- In vivo and ex vivo assessment of puncture performance
- Longitudinal monitoring of needle and skin dynamics
- Non-invasive imaging suitable for repeat measurements
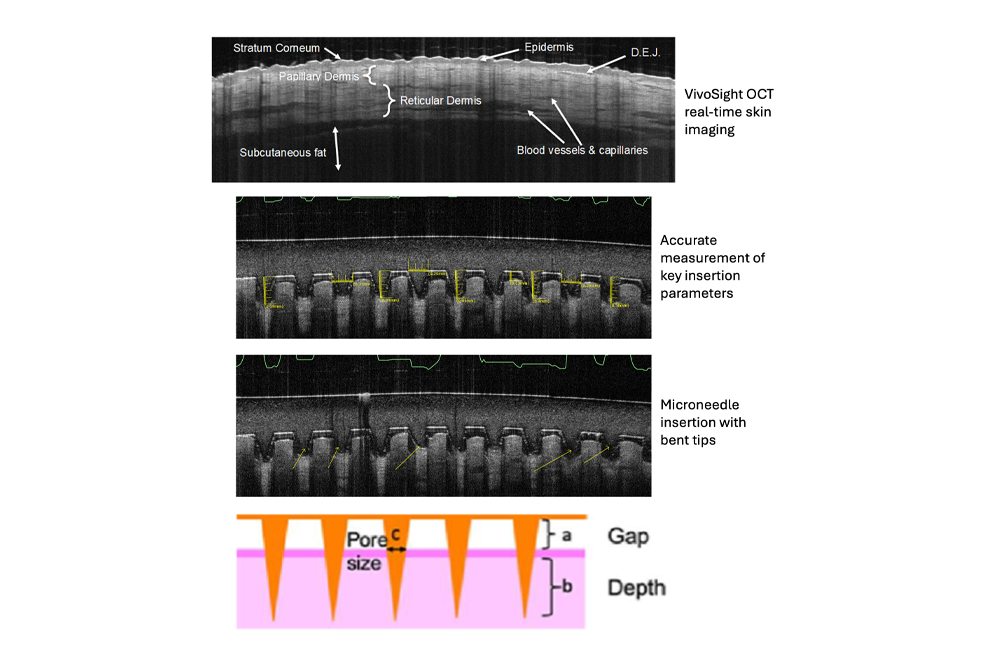
Visualise and quantify key MAP penetration metrics – in vivo
Quantify critical MAP performance parameters
VivoSight transforms microneedle imaging into objective, quantitative data that supports device optimisation, formulation decisions and regulatory evidence generation.
Measure and monitor:
- Needle dimensions and geometry
- Penetration and insertion depth
- Dissolution and swelling behaviour
- Pore diameter and air gap
- Skin defect kinetics and recovery
Measurements can be captured immediately after application and over extended time periods, enabling detailed assessment of MAP performance and consistency.
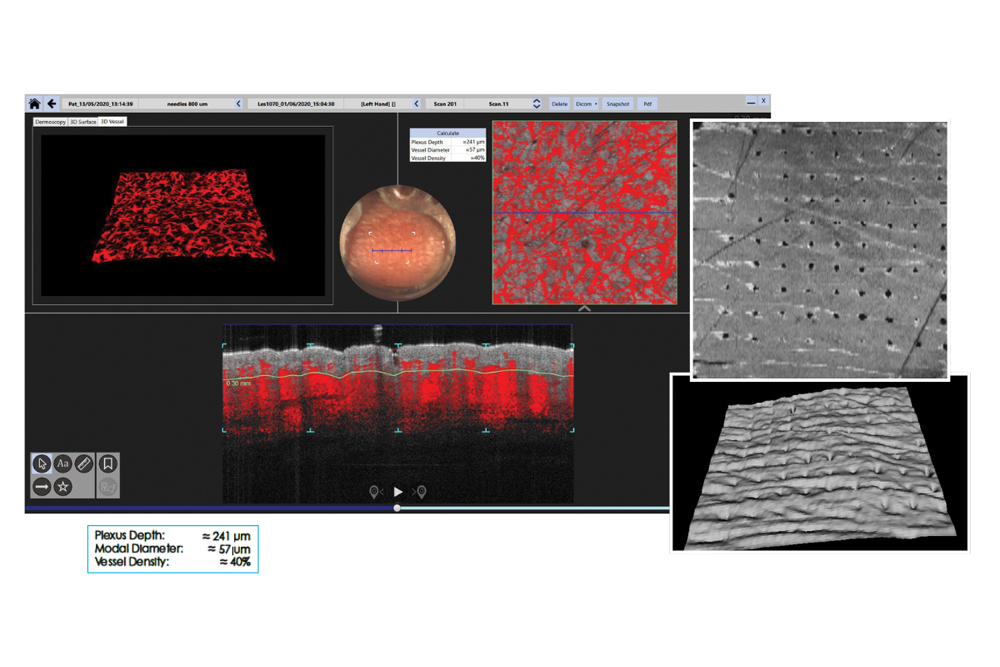
Visualise vascular response and skin surface dynamics

See microneedle–skin interaction in vivo in real time
Generate quantitative evidence of puncture performance, dissolution and skin recovery without tissue sectioning.

Monitor needle dynamics and skin response over time
VivoSight allows continuous or repeated imaging over time, supporting dynamic MAP studies that are difficult or impossible with destructive techniques.
- Monitoring microneedle dissolution and swelling for up to 60 minutes in vivo
- Visualising vascular changes induced by MAP insertion
- Assessing inflammatory response and skin recovery post-removal
- Documenting hole closure and longer-term skin effects

Continuous in-vivo imaging of MAP behaviour
Faster, more cost-effective MAP development
By eliminating the need for routine histology and sectioning, VivoSight significantly reduces development timelines and costs while increasing the volume and quality of data collected.
Benefits include:
- Rapid, repeatable measurements from a single sample
- Reduced reliance on destructive ex vivo analysis
- Improved study efficiency and throughput
- Strong visual and quantitative evidence to support development and regulatory discussions

In vivo and ex vivo flexibility
VivoSight supports both in vivo clinical imaging and high-definition ex vivo analysis.
For laboratory workflows, the optional 3-axis Probe Stand converts the handheld VivoSight probe into a stable OCT microscope, enabling fast, precise imaging of excised skin samples while maintaining the same imaging modality used in vivo.
Designed for clinical and research environments
- Device cleared for use in clinical settings
- Non-invasive, patient-friendly imaging
- Suitable for academic, industrial and translational MAP research
Transform your MAP
research with VivoSight
Accelerate decisions. Reduce development time. Strengthen your data.
From early-stage formulation development to late-stage optimisation and commercialisation, VivoSight Dx Pro delivers objective, repeatable evidence of microneedle–skin interaction — in vivo and in real time.
